NF-kappa-B inhibitor alpha NF-kappa-B inhibitor alpha ikB-alpha IkappaBalpha I-kappa-B-alpha nuclear factor of kappa light chain gene enhancer in B-cells major histocompatibility complex enhancer-binding protein MAD3
基本介紹
- 中文名:人核因子κB抑制蛋白α
- 外文名:IKB-α
名稱,基因蛋白信息,核因子,蛋白家族,研究進展,
名稱
Preferred Names:
NF-kappa-B inhibitor alpha
NF-kappa-B inhibitor alpha
ikB-alpha
IkappaBalpha
I-kappa-B-alpha
nuclear factor of kappa light chain gene enhancer in B-cells
major histocompatibility complex enhancer-binding protein MAD3
基因蛋白信息
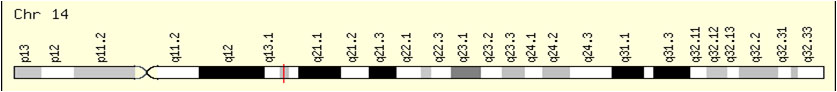
IKB-α的 基因定位於14號染色體長臂1區3亞區的2號位上
Summary:This gene encodes a member of the NF-kappa-B inhibitor family, which contain multiple ankrin repeat domains. The encoded protein interacts with REL dimers to inhibit NF-kappa-B/REL complexes which are involved in inflammatory responses. The encoded protein moves between the cytoplasm and the nucleus via a nuclear localization signal and CRM1-mediated nuclear export. Mutations in this gene have been found in ectodermal dysplasia anhidrotic with T-cell immunodeficiency autosomal dominant disease.
Protein function:Inhibits the activity of dimeric NF-kappa-B/REL complexes by trapping REL dimers in the cytoplasm through masking of their nuclear localization signals. On cellular stimulation by immune and proinflammatory responses, becomes phosphorylated promoting ubiquitination and degradation, enabling the dimeric RELA to translocate to the nucleus and activate transcription.
蛋白序列:
translation="MFQAAERPQEWAMEGPRDGLKKERLLDDRHDSGLDSMKDEEYEQ
MVKELQEIRLEPQEVPRGSEPWKQQLTEDGDSFLHLAIIHEEKALTMEVIRQVKGDLA
FLNFQNNLQQTPLHLAVITNQPEIAEALLGAGCDPELRDFRGNTPLHLACEQGCLASV
GVLTQSCTTPHLHSILKATNYNGHTCLHLASIHGYLGIVELLVSLGADVNAQEPCNGR
TALHLAVDLQNPDLVSLLLKCGADVNRVTYQGYSPYQLTWGRPSTRIQQQLGQLTLEN
LQMLPESEDEESYDTESEFTEFTEDELPYDDCVFGGQRLTL"
核因子
核因子-KB(nuclear factor-kappa B,NF-KB)蛋白家族是一種多效性的轉錄因子,可以與多種基啟動子部位的KB位點發生特異性的結合從而促進其轉錄表達。其受氧化應激、細菌脂多糖,細胞因子等多種刺激而活化後,能調控前炎症性細胞因子、細胞表面受體、轉錄因子、粘附分子等的生成。而這些刺激因素及其調控的因子與微循環障礙的發生、發展均有著密切的關係。
蛋白家族
IKB蛋白家族成員有IKBα(MAD-3,pp40)、IKBβ、IKBγ/p105、IKBδ/p100、IKBε、Bcl-3以及果蠅屬的Cactus等。其家族結構特點是均有多個約33個胺基酸的重複序列,稱為崐SWI6/錨蛋白重複序列,主要參與與Rel蛋白的RHD相互作用。
IKB蛋白主要有以下三個部分構成:
1.與蛋白降解有關的N-末端區;
2.能與NF-KB相互作用的內部區(區內含有錨蛋白重複序列ANK)(家族共有結構)主要參與與Rel蛋白的RHD相互作用;
3.稱為PEST的C-端區,主要參與“囚禁”NF-KB在細胞漿中。
IKB蛋白主要有以下三個部分構成:
1.與蛋白降解有關的N-末端區;
2.能與NF-KB相互作用的內部區(區內含有錨蛋白重複序列ANK)(家族共有結構)主要參與與Rel蛋白的RHD相互作用;
3.稱為PEST的C-端區,主要參與“囚禁”NF-KB在細胞漿中。
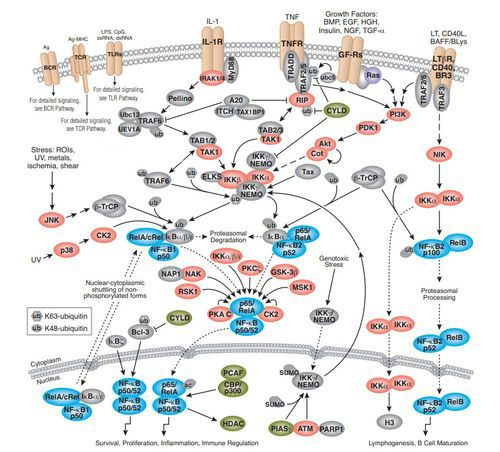
IKBs家族參與兩個信號通路
NF-kB signal pathway
NF—KB存在於多種細胞內, 是一種具有多向性調節作用的核轉錄因子,參與炎 症、免疫、細胞增殖和細胞凋亡等多種生理、病理過 程的基因調控。
NF-KB系統由NF—KB家族及其抑制 物IKB家族共同組成。前者由Rel蛋白家族成員構 成,以同源或異源二聚體的形式存在。哺乳動物細 胞中有5種NF—KB/Rel家族成員:RelA(p65)、RelB、 C-Rel、p50(NF-KB1)和p52(NF-KB2),其N端均含 有一段保守的約300個胺基酸殘基的Rel同源結構 域,該結構域包含Rel蛋白間聚合位點及與IKB結合 的位點,同時也包含DNA識別序列和核定位序列。 只有NF-KB蛋白二聚體才具有DNA結合活性,不同 NF-KB/Rel蛋白二聚體具有不同的結合序列,識別不 同的DNA靶目標,從而調節不同基因的表達。
NF-KB的激活必須通過兩個步驟:①IKB從NF-KB復 合體中解離並降解,暴露NF-KB的核定位序列; ②NF-KB發生核易位,並與特異的KB序列結合。
經典途徑:某些配體(TNF、IL-1、Lsp、CD 40L等)與細胞表面相應受 體結合通過胞質側結構域募集到一些接頭蛋白可以 激活經典途徑,這些接頭蛋白能夠募集並激活IKB 激酶複合物(inhibitor of kappa B kinase complex,IKK),激活 後的IKK複合體將IKB的32和36位兩個保守的絲 氨酸殘基磷酸化,磷酸化的IKB發生多泛素化,最終 由26S蛋白體降解,被釋放的NF-KB發生核易位,作用於相應的靶基因。
非經典途徑:與經典途徑不同的是:信號相 關接頭蛋白只能被一些有限的信號分子,如淋巴細 胞毒素B與CD40L激活,且並不需要泛素蛋白的參 與。信號分子與受體的結合將激活NF-KB誘導激 酶,活化的激酶磷酸化IKK-α,繼而將p100磷酸化, 磷酸化的p1O0部分水解後轉變為p52,其核定位信 號和DNA結合結構域暴露,可與Rel-B組成複合體 進人胞核激活靶位點。
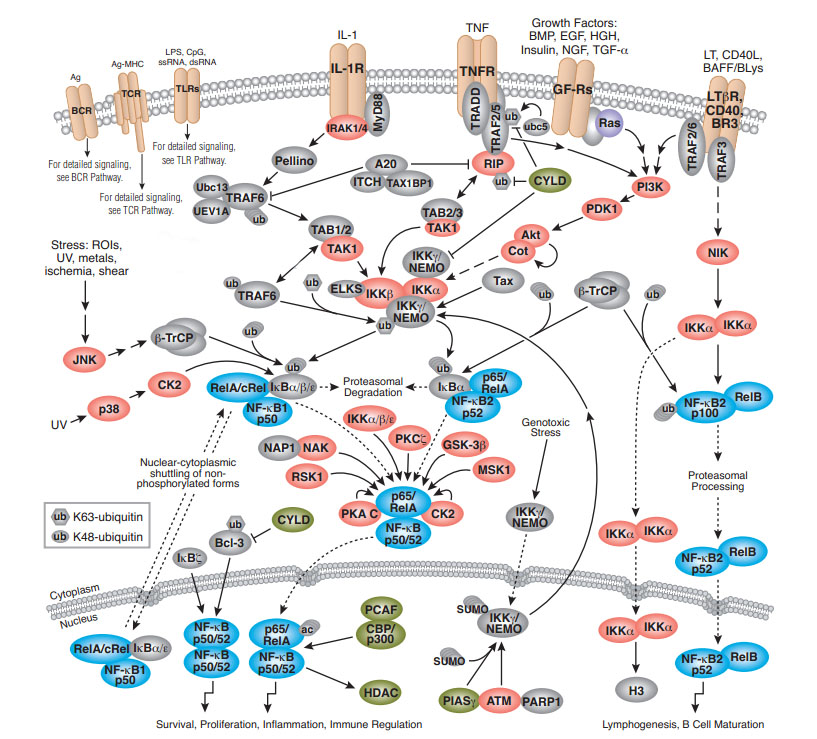
Toll-like Receptor signal pathway
類Toll受體(Toll-like receptors, TLR) 是最古老最保守的免疫系統的組成部分。所以Toll-like receptor信號通路主要作用是調控一些參與免疫的蛋白表達。
TLR多表達於哺乳動物的抗原提呈細胞以及上皮細胞, 被認為是具有相同結構的I型跨膜型模式識別受體(pattern recognition receptors, PRR) 。
PRR由緊密相連的三個部分組成:
1、胞外段是富含亮氨酸重複序列(leucine2richrepeat , LRR)的N端,約550~980個胺基酸組成;
2、跨膜區是富含半胱氨酸的結構域;
3、 胞內段與IL21受體同源, 被稱為TIR(Toll/ IL21R) , 約200個胺基酸大小。LRR結構域是所有PRR共有的結構特點, 它識別廣泛存在於病原體細胞表面的分子標誌, 如酵母細胞壁表面上的甘露糖, 以及脂多糖、多肽糖、胞壁酸等各種細菌的細胞壁成分等,這些分子被統稱為病原相關分子模式(pathogen-associated molecular patterns,PAMP) 。兩者結合啟動信號轉導過程。TIR結構域在細胞內介導信號傳遞, 在蛋白之間相互作用過程中意義重大, 與天然免疫和獲得性免疫作用的發揮緊密相連
lTLRs家族有13個蛋白,TLR1-9為人鼠共有,TLR10人獨有,TLR11-13鼠獨有。其中TLR1,TLR2,TLR4,TLR5,TLR6位於細胞表面,TLR3,TLR7,TLR8,TLR9是定位於核內體/溶酶體。
研究進展
Novel 2-Phenyl-1-Pyridin-2yl-Ethanone (PpY) Based Iron Chelators Increase Expression of IkBα and Heme Oxygenase-1 and Inhibit HIV-1
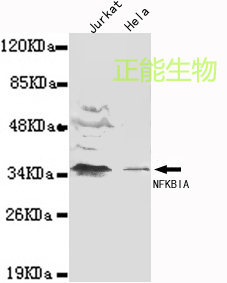
HIV-1 transcription is activated by HIV-1 Tat protein, which recruits CDK9/cyclin T1 and other host transcriptional co-activators to the HIV-1 promoter. Tat itself is phosphorylated by cell cycle kinase 2 (CDK2) and inhibition of CDK2 by small interfering RNA or iron chelators inhibits HIV-1 transcription. HIV-1 transcription is also activated by NF-kB that binds to HIV-1 LTR independent to Tat but can also be recruited Tat-dependently by CDK9/cyclin T1. Recently, induction of heme oxygenase-1 (HO-1) by hemin was shown to inhibit HIV-1 in vitro and in vivo. Here, we analyzed the effect of novel phenyl-1-pyridin-2yl-ethanone (PPY) based iron chelators, PPYeT and PPYaT, on HIV-1. Both chelators efficiently inhibited one round of HIV-1 replication in T cells at low nanomolar IC50s without exhibiting cytotoxicity at 24 hrs incubation. The iron chelators efficiently bound intracellular labile iron as it was determined in calcein binding assays. Because we previously showed that iron chelators inhibited the activity of CDK9, we analyzed expression of several cellular genes dependent on CDK9. Unexpectedly, chelators were found to induce the expression of IkBα, an inhibitor of NF-kB (Fig1). Treatment with the iron chelators retained NF-kB in cytoplasm of the treated cells suggesting reduction in NF-kB in nucleus (Fig2). The chelators were also shown to induce HO-1 expression in cultured monocytes, likely to do a decrease of intracellular iron pool. This effect of iron chelators mimicked the effect of hemin treatment which also induced HO-1 and inhibited HIV-1 infection in our experimental conditions. Low nanomolar IC50s for the PPY-based iron chelators and lack of toxicity suggest their potential usefulness as future anti-retroviral therapeutics. Further studies are needed to investigate additional targets for iron chelators in HIV-1 life cycle that may include reverse transcription and capsid assembly. Therefore iron chelators need to be carefully assessed not only to understand the mechanism but also as a therapeutic strategy.